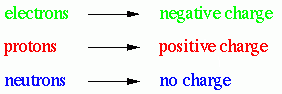
All matter is made up of tiny objects called
atom.
Atoms have three
different components -
protons,
neutrons
and
electrons
Each of these
components have different properties and one of these properties is their
charge.

|
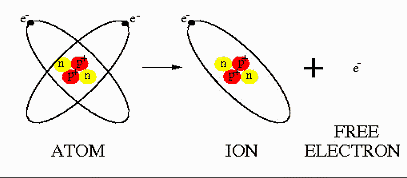
In atoms, the protons and neutrons lie in the centre of the atom
called the nucleus and around
the nucleus lie the electrons. There are equal numbers of protons and
electrons in atoms so an atom is electrically neutral. Sometimes when
the electrons gain energy they can escape from the atom leaving a free
negative charge and a positively charged atom called an
ion.
This is where our positive and negative electric charges
come from.
|


|